|
Bioadhesion is broad and complex topic. We divide our efforts, in bioadhesion, into the investigation of antifouling technologies as well as “fouling” technologies. Antifouling is focused on the prevention of the undesired adhesion of materials and organisms to surfaces. These surfaces are primarily exposed to marine environments, but also are implanted in the human body or used in other environments. We have recently developed the Engineered Roughness Index model that illustrates the importance of the surface microtopography on cell adhesion to our surfaces. The model is:
|
|
The terms are based upon the interaction between Wenzel theory of wetting and the Cassie-Baxter theory. The intersection of these two concepts occurs at a critical angle for a spreading liquid front on a microtopography. We have demonstrated that the ERI Model works very well for the inhibition of adhesion, but we have focused our efforts recently on the investigation of prokaryotic cells on surfaces. We continue refining our model as we obtained more data on organisms other than marine organisms as well as other biocompatible materials such as our resorbable bioactive materials.
Contact Guidance:
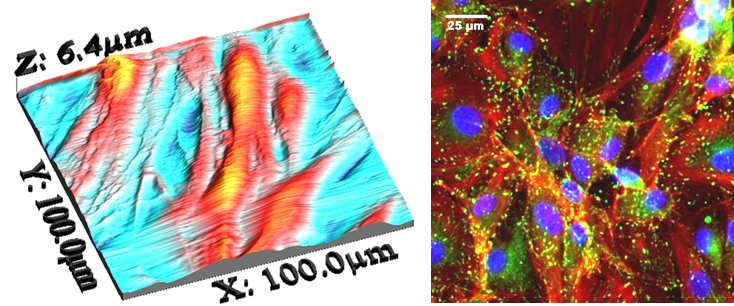
A phenomenal, observed in 1914 by Harris, illustrates the importance of topography on cell adhesion. Harris’ work is the basis for much of the current research on the effects of structure on tissue engineering. Cells follow the contours of surfaces in different ways as the scale varies. The slide below illustrates the behavior of Porcine Cardiovascular Cells on silicone elastomers with channels 10 micons square. The channels induce highly elongated phenotypes. The sections between the channels enable the cells to adapt to their normal physiological forms.
We have also demonstrated the effects of our microtopographies on both human endothelial progenitor and human smooth muscle cell progenitor cells.